
Mass increases as aqueous ions turn to solid at the cathode. The nature of the cathode rays depends on the nature of the gas filled inside the discharge tube. In the cathode ray tube experiment, the gas is maintained at a very low pressure up to 10−3 mm Hg, so that there is enough space for electrons to accelerate before colliding with more atoms, ejecting a large number of free electrons. He concluded that the atom can be divided into smaller particles. The characteristics of these rays do not depend upon the nature of the electrode material or the nature of the gas-filled tube. Thomson concluded that the rays were negatively charged and moved around the positive charge. What were three major conclusions of Thomson’s cathode ray tube experiment? This proved that the negative charge and the ray were inseparable and intertwined. Thomson’s First Cathode Ray Experiment He found that by applying a magnetic field across the tube, there was no activity recorded by the electrometers and so the charge had been bent away by the magnet.  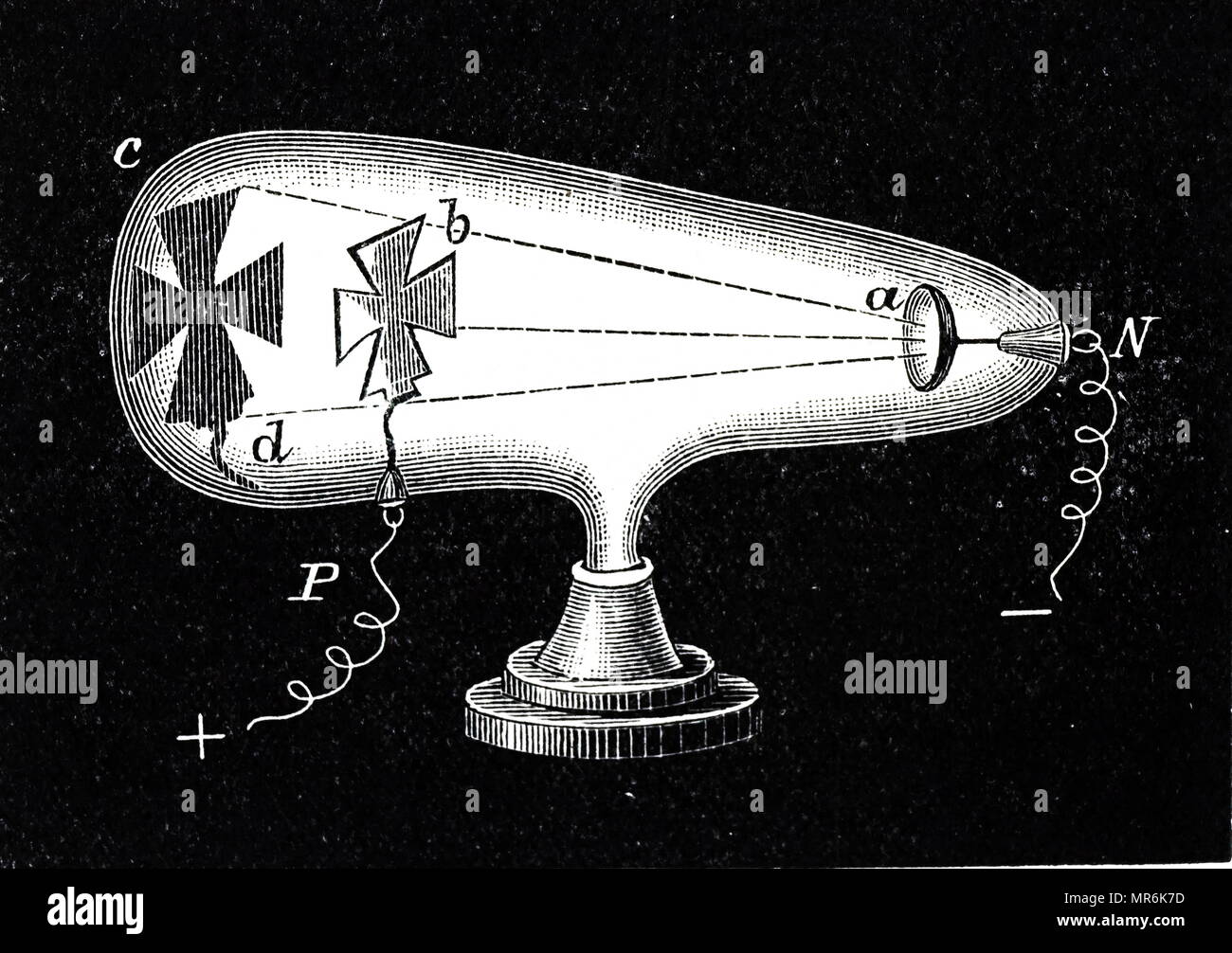
How does the cathode ray tube experiment proved the existence of electrons? This theory further helped physicists in understanding the structure of an atom. Thomson concluded that rays were and are basically negatively charged particles present or moving around in a set of a positive charge. What was the conclusion of the cathode ray experiment?Ĭonclusion. They travel with a velocity of upto one tenth of velocity of light and they effect photographic plates. What is the velocity of cathode rays?Ĭathode rays are stream of electrons observed in vacuum tubes and travel in straight lines. Type III: Make the cathode between ¾ – 1 times the max anode area, about 1:1.3, up to 1:1. Type II: Make the cathode about ½ the max anode surface area: 1:2, cathode to anode. What is the ratio of cathode and anode?Ĭathode to anode recommendations: A ratio of 1:10 is often used. It is independent of the nature of gas in discharge tube and nature of cathode or electrodes of tube. The e/m ratio for cathode rays is constant. Property 4: They are produced at the cathode and travel to the anode in a vacuum tube. Property 3: Electric and magnetic fields deflect cathode rays. Property 1: Cathode rays travel in a straight line and they can cast sharp shadows. What are the 4 properties of cathode rays? The cathode ray tube was invented by Sir William Crookes. This occurs because electrons are negatively charged particles, and are therefore repelled by negative charge and attracted to positive charge. In a cathode ray tube, electrons move in a beam from negative end (cathode) of the tube to the positive end (anode). How do particles move in a cathode ray tube? Thomson installed a set of parallel plates attached to a variable voltage supply. Therefore the cathode rays must have a very small mass. The path of the cathode rays is straight and horizontal, which indicates that gravity has very little effect. 
Thomson proposed the plum pudding model of the atom, which had negatively-charged electrons embedded within a positively-charged “soup.” Why are cathode rays not affected by gravity? Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. What was the hypothesis of the cathode ray tube experiment? 24 Why anode is bigger than cathode? How do you calculate cathode rays?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
